 Therefore, you can predict that bromine, #"Br"_2#, which is the biggest molecule of the group, will exhibit the strongest London dispersion forces and thus have the highest boiling point.īy the same logic, fluorine, #"Fl"_2#, which is the smallest molecule of the group, will exhibit the weakest London dispersion forces and thus have the lowest boiling point. Think of it like this - the bigger the surface are and the magnitude of the partial charges, the stronger the atraction. An example of this would be neopentane - C (CH3)4 - which has a boiling point of 282. 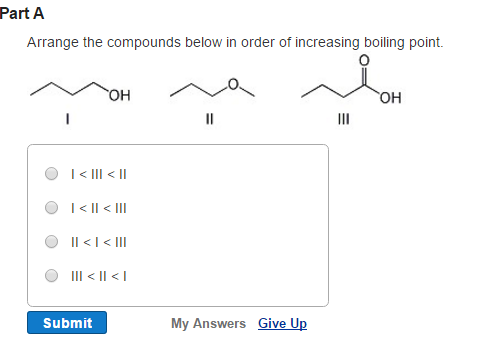
This means that the instantenous distorsions in the electron distribution will create more poweful temporary dipoles. Branching of carbon compounds have lower boiling points. Now, as you go down the group, the size of the atoms, and implicitly the size of their electron clouds, increase. 
This means that the halogen molecule that exhibits the strongest London dispersion forces will have the highest boiling point. This means that for very short period of time, the two molecules are attracted to each other.Īs you know, the stronger the intermolecular forces of attraction, the higher th boiling point. it induces a similar distorsion in the second molecule's electron cloud. Hydrogen bonding is more stronger in nature as compared to a molecule with dipole-dipole interactions. London Forces and Their Effects Order the following compounds of a group 14 element and hydrogen from lowest to highest boiling point: CH4. Therefore, higher will be its boiling point. When a molecule that exhibits such dipole moment comes near another molecule, it polarizes its electron cloud, i.e. Also, more is the electronegative atom attached to the hydrocarbon more is the partial charges on the atoms of a molecule. It is the weakest intermolecular force of attraction, therefore, the v apor pressure will be. The vapor pressure of the compounds can be explained as: Bromine is a gaseous compound, which has the Van der Waal force of attraction between its molecules. This leads to the creation of a temporary dipole moment. The compounds arranged in the decreasing order of their vapor pressure are: Br HO NCl. Likewise, regions with lower electron density exhibit partial positive charges. In regions with higher electron density, partial negative charges arise. These distorsions take place when instantaneous changes in how the electrons are distributed in the molecule's electron cloud lead to regions that have a higher electron density and regions that have a lower electron density. 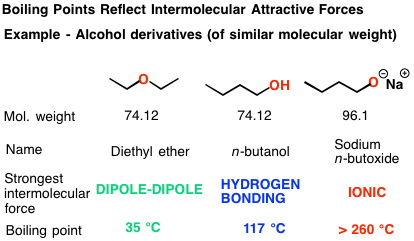
Now, London dispersion forces are actually caused by instantaneous and random distorsions in a molecule's electron cloud. Since these molecules are nonpolar, they will only exhibit weak London dispersion forces. The halogens, which are the lements that make up group 17 of the periodic table, exist as diatomic molecules. In your case, you have to find how the boling points of three nonpolar molecules relate to each other. The expected valence for the dbr and e/o calculations is given in the. As you know, a molecule's boiling point depends on the strength of the intermolecular forces of attraction its molecules exhibit. This Body Fat Navy Calculator uses the U. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
